Award for excellent research in cell biology
Dr. Andrew Clark (Institute of Cell Biology and Immunology/Stuttgart Research Center Systems Biology) was recently awarded the Walter Flemming Prize by the German Society for Cell Biology (DGZ). This award is named after a German cell biologist who made fundamental contributions to our understanding of cell division and replication. The Walter Flemming Prize is awarded annually to an experienced postdoctoral researcher or early research group leader for excellent research in the field of cell biology.
Dr. Clark received the Walter Flemming Award for his contribution to our understanding of collective cell migration and the mechanobiology of cells and tissues. Collective cell migration is a key biological process involved in development, tissue repair, and cancer. In invasive cancer, tumor cells actively migrate away from the primary tumor and eventually reach other organs, where they colonize and form new tumors. This process, called metastasis, is responsible for about 90% of cancer-related deaths. In recent years, it has become increasingly clear that this migration process can be controlled by changes in the mechanical properties of tumor cells and the tissue surrounding the tumor.
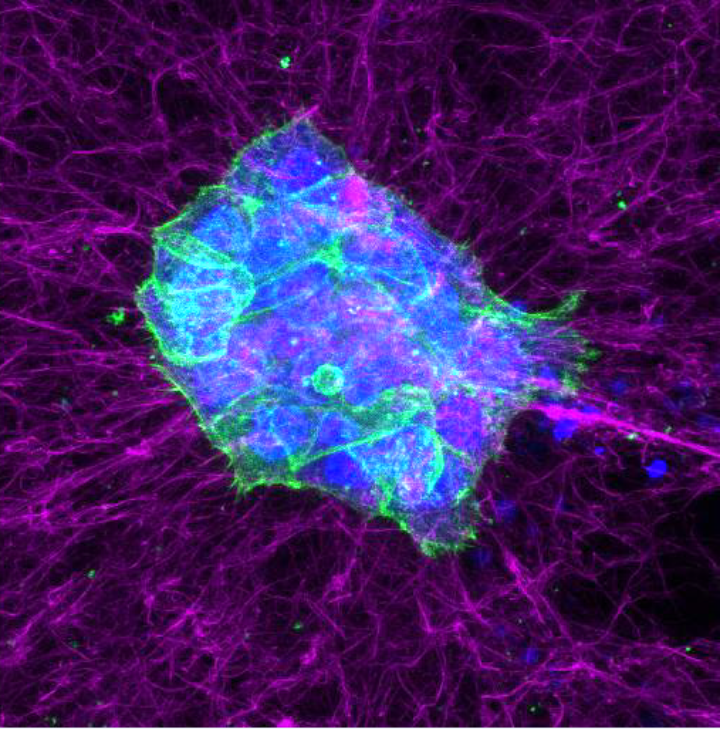
During his doctoral research at the Max Planck Institute of Molecular Cell Biology and Genetics in Dresden, Dr. Clark discovered new mechanisms underlying the organization and regulation of the actin cortex. The actin cortex is a cytoskeletal network that is largely responsible for controlling cell surface mechanics and cell shape changes during processes such as cell migration and cell division. His findings demonstrated for the first time that the architecture of this network is crucial for the control of cell mechanics.
After successfully completing his doctoral degree studies, Dr. Clark went to Paris and worked as a postdoctoral fellow at the Institut Curie in the laboratory of Danijela Matic Vignjevic. During this time, he studied collective cell migration in various contexts, including cancer cell migration in intestinal tumors. During his postdoc, he discovered new mechanisms of mechanical feedbacks between collectively migrating cell clusters and collagen networks that make up the connective tissue in many organs and surrounding tumors. This coupling, which depends on the mechanical properties of the collagen network, promotes the sustained collective migration of cancer cell clusters. In the future, these findings could be applied to the diagnosis of cancer metastasis and also inspire new therapeutic approaches that target the mechanistic interactions between cancer cells and their microenvironment.
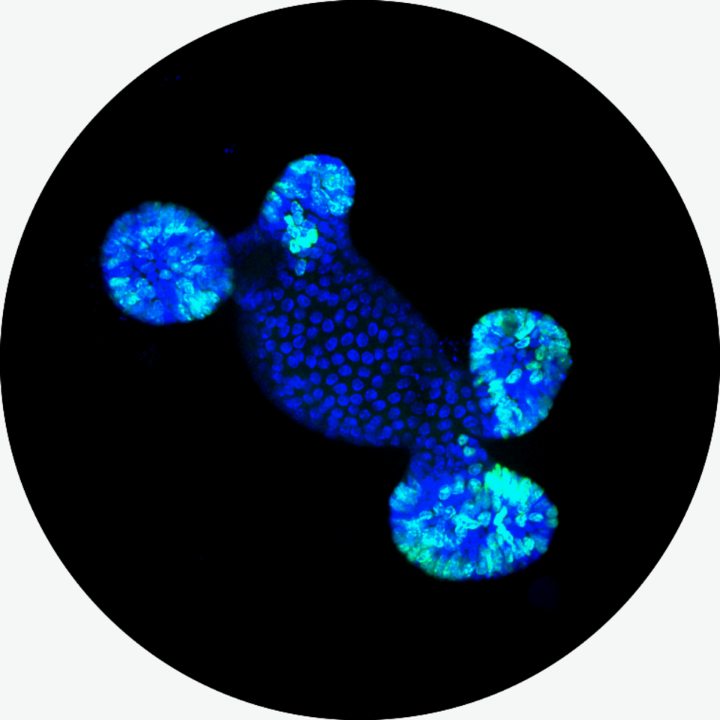
Andrew started his own research group in May 2021 as a joint junior research group at the University of Stuttgart, Institute of Cell Biology and Immunology/Stuttgart Research Center Systems Biology and the University of Tübingen, Center for Personalized Medicine. His research group studies the collective migration and tissue architecture of the small intestine in physiology and disease, primarily using primary intestinal organoids ("mini-intestines").
Selected publications
Clark, A.G., Maitra, A., Jacques, C., Bergert, M., Pérez-González, C., Simon, A., Lederer, L., Diz-Muñoz, A., Trepat, X., Voituriez, R. and Vignjevic D.M. (2022) Self-generated gradients steer collective migration on viscoelastic collagen networks. Nature Materials. 21:1200-1210. https://doi.org/10.1038/s41563-022-01259-5
Staneva, R., El Marjou, F., Barbazan, J., Krndija, D., Richon, S., Clark, A.G.*, and Vignjevic, D.M.* (2019) Cancer Cells in the Tumor Core Exhibit Spatially Coordinated Migration Patterns. Journal of Cell Science, 132(6):jcs220277. https://doi.org/10.1242/jcs.220277
Chugh, P.*, Clark, A.G.*, Smith, M.B.*, Cassani, D.A.D., Ragab, A., Roux, P.P., Charras, G., Salbreux, G. and Paluch, E.K. (2017) Actin Cortex Architecture Regulates Cell Surface Tension. Nature Cell Biology, 19(6):689-697. https://dx.doi.org/10.1038/ncb3525
Clark A.G., Dierkes K. and E.K. Paluch (2013) Monitoring Actin Cortex Thickness in Live Cells. Biophysical Journal. 105(3):570-580. https://doi.org/10.1016/j.bpj.2013.05.057