Development of Recycling Strategies for All-solid-state Batteries
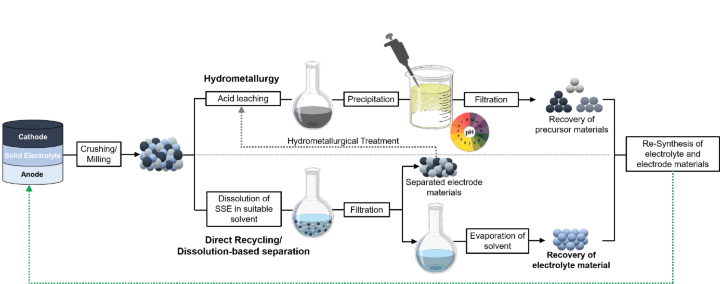
All-solid-state batteries (ASSBs) with solid-state electrolytes (SSEs) are promising candidates for future battery technologies. Despite the tremendous efforts made on a performance-oriented pathway, the recycling of such batteries, thus the sustainability of several critical elements (i.e., lithium, rare earth, cobalt, …) implemented in such batteries, are less studied by the scientific community. Our group is currently developing recycling strategies for ASSBs with different solid-state electrolyte classes (e.g., oxides (i.e., Li7La3Zr2O12 (LLZO) [1,2]) sulfides (i.e., ß- Li3PS4 [3]) and halides (i.e., Li3InCl6)) in combination with different conventional electrode materials (e.g., Li4Ti5O12 (LTO), LiNixMnyCozO2 (NMC), LiCoO2 (LCO), LiMn2O4 (LMO), LiFePO4 (LFP), LiNixCoyAlzO2 (NCA)). Our studies indicate that processes should be individually adapted to a specific SSE class [4]. ASSBs with oxide- based SSEs are preferably treated via hydrometallurgical methods (Figure 1, above), whereas the direct recycling/ dissolution-based separation method (Figure 1, down) is principally applicable for sulfide- and halide- based SSEs.
[1] M. A. Nowroozi, A. I. Waidha, M. Jacob, P. A. van Aken, F. Predel, W. Ensinger, O. Clemens, ChemistryOpen 2022, 11, e202100274.
[2] A. I. Waidha, A. Salihovic, M. Jacob, V. Vanita, B. Aktekin, K. Brix, R. Kautenburger, J. Janek, W. Ensinger, O. Clemens, ChemSusChem 2023, 16, e202202361.
[3] K. Wissel, L. M. Riegger, C. Schneider, A. I. Waidha, T. Famprikis, Y. Ikeda, B. Grabowski, R.E. Dinnebier, B.V. Lotsch, J. Janek, W. Ensinger, O. Clemens, ACS Applied Energy Materials 2023, 6, 7790-7802.
[4] M. Jacob, K. Wissel, O. Clemens, Materials Futures 2023.
Intercalation based electrode materials for Fluoride Ion Batteries
Fluoride Ion Batteries present a valid strategy to exploit alternative ressources, making future energy technologies less dependent on key elements, e. g., Li and Co. Those batteries work based on a fluoride ion shuttle. Our group is developing intercalation based compounds as active electrode materials, where we investigated anion-deficient perovskites [1], schafarzikite type [2] as well as Ruddlesden-Popper type compounds [3-5], where the latter have shown to possess superior cycling stability. Currently, we focus on developing those materials further, aiming to understand interface stabilities and trying to develop strategies to stabilize the crucial carbon additive – active electrode material interfaces at both, the cathode and anode side.
[1] O. Clemens, C. Rongeat, M. A. Reddy, A. Giehr, M. Fichtner, H. Hahn, Dalton Transactions 2014, 43, 15771-15778.
[2] M. A. Nowroozi, B. de Laune, O. Clemens, ChemistryOpen 2018, 7, 617-623.
[3] M. A. Nowroozi, K. Wissel, J. Rohrer, A. R. Munnangi, O. Clemens, Chemistry of Materials 2017, 29, 3441-3453.
4] M. A. Nowroozi, O. Clemens, ACS Applied Energy Materials 2018, 1, 6626-6637.
[5] M. A. Nowroozi, S. Ivlev, J. Rohrer, O. Clemens, Journal of Materials Chemistry A 2018, 6, 4658-4669.
Topochemical Fluorination and Defluorination of Oxide Materials – Targeting Material Properties
Perovskite and perovskite related materials are known for their various properties, among them magnetic, electrical and catalytic properties. Those properties are often connected to the detailed transition metal oxidation state as well as structural arrangement.
In our group we investigate ways to make new compounds and to study them for their resulting properties. One such route was found in combining subsequent topochemical substitutive fluorination (1 oxide for 2 fluoride ions) [1-3] and reductive defluorination, which has provided a way to prepare new reduced titanates at temperatures as low as 300 °C [4], and we have now extended our studies to target the structural and magnetic properties of Ni-based Ruddlesden-Popper type phases [5].
In addition, we aim to exploit electrochemical fluorination/defluorination reactions within Fluoride Ion Battery cells to reversibly change magnetic properties of this material class, which cannot be reversibly targeted by alkali ion batteries (lithium or sodium) otherwise.
[1] O. Clemens, F. J. Berry, A. J. Wright, K. S. Knight, J. M. Perez-Mato, J. M. Igartua, P. R. Slater, Journal of Solid State Chemistry 2013, 206, 158-169.
[2] O. Clemens, P. R. Slater, Reviews in Inorganic Chemistry 2013, 33, 105-117.
[3] O. Clemens, R. Kruk, E. A. Patterson, C. Loho, C. Reitz, A. J. Wright, K. S. Knight, H. Hahn, P. R. Slater, Inorganic Chemistry 2014, 53, 12572-12583.
[4] K. Wissel, S. Dasgupta, A. Benes, R. Schoch, M. Bauer, R. Witte, A. D. Fortes, E. Erdem, J. Rohrer, O. Clemens, Journal of Materials Chemistry A 2018, 6, 22013-22026.
[5] K. Wissel, J. Heldt, P. B. Groszewicz, S. Dasgupta, H. Breitzke, M. Donzelli, A. I. Waidha, A. D. Fortes, J. Rohrer, P. R. Slater, G. Buntkowsky, O. Clemens, Inorganic Chemistry 2018, 57, 6549-6560.
Topochemical modifications of thin film compounds
Many fluorinated perovskite or perovskite-related oxides are metastable. Therefore, it is difficult to study bulk material properties (especially electrical properties), which require the availability of dense ceramics. The same hold true for hydrated perovskite phases, which cannot be sintered in the hydrated phase, and where volume changes on hydration are often far too high to allow for a destruction free hydration of dense ceramics.
Therefore, we have used methods to topochemically fluorinate [1] and hydrate [2,3] epitaxially grown thin films. These can then be studied as a model system representing the grain-boundary free, single-crystalline state in order to understand electrical and magnetic properties of the bulk state in a more profound way. In addition, a combination of strain engineering [4] and topochemical reactions might serve to induce properties which deviate from the unstrained state.
[1] P. A. Sukkurji, A. Molinari, C. Reitz, R. Witte, C. Kuebel, V. S. K. Chakravadhanula, R. Kruk, O. Clemens, Materials 2018, 11, 1204.
[2] P. A. Sukkurji, A. Molinari, A. Benes, C. Loho, V. S. K. Chakravadhanula, S. K. Garlapati, R. Kruk, O. Clemens, Journal of Physics D-Applied Physics 2017, 50, 115302.
[3] A. Benes, A. Molinari, R. Witte, R. Kruk, J. Broetz, R. Chellali, H. Hahn, O. Clemens, Materials 2018, 11, 52.
[4] O. Clemens, N. Kunkel, Nachrichten aus der Chemie 2019, 67, 59-62.
New proton and electron conducting perovskites for protonic ceramic fuel cells
Whereas water uptake is well known for Ba-rich perovskites such as BaInO2.5 and BaZr1-xYxO3-x/2, redox-active, anion deficient ferrates and cobaltates have not been studied for large water uptakes [1,2]. Only recently, our group has prepared high water content barium ferrates and cobaltates [3,4], which show strong relaxation around the incorporated protons. Those were found to induce proton conductivity in such systems, which makes them interesting for reduced temperature electrode (300 – 600 °C) catalysts within protonic ceramic fuel cells
In this context, we combine studies of variable temperature X-ray and neutron diffraction, thermal analysis, impedance spectroscopy as well as FT-IR to gain understanding of the factors which help to stabilize protonic charge carriers in a Ba-rich perovskite matrix. Recently, we have moved to understand their catalytic properties for alkaline fuel cells in addition.
[1] O. Clemens, M. Groeting, R. Witte, J. Manuel Perez-Mato, C. Loho, F. J. Berry, R. Kruk, K. S. Knight, A. J. Wright, H. Hahn, P. R. Slater, Inorganic Chemistry 2014, 53, 5911-5921.
[2] A. I. Waidha, H. Zhang, M. Lepple, S. Dasgupta, L. Alff, P. Slater, A. D. Fortes, O. Clemens, Chem Commun 2019, 55, 2920-2923.
[3] P. L. Knoechel, P. J. Keenan, C. Loho, C. Reitz, R. Witte, K. S. Knight, A. J. Wright, H. Hahn, P. R. Slater, O. Clemens, Journal of Materials Chemistry a 2016, 4, 3415-3430.
[4] A. I. Waidha, M. Lepple, K. Wissel, A. Benes, S. Wollstadt, P. R. Slater, A. D. Fortes, O. Clemens, Dalton Transactions 2018, 47, 11136-11145.
Further topics
Magnetic properties of manganese vanadates
[1] O. Clemens, A.J. Wright, K.S. Knight, P.R. Slater, Dalton Trans. 42 (2013) 7894-7900.
2] O. Clemens, J. Rohrer, G. Nenert, Dalton Trans. 45 (2016) 156-171.